And the Nobel Prize in Geology Goes to…
by John Weidner, MSDC Treasurer
Oh, wait! There is no Nobel Prize in geology. But if there were, Norman Bowen would surely have won it for his discovery of Bowen’s Reaction Series.

Many people think the Nobel Prizes are so named because the achievements are wonderful, noble, worthy. Nope! It's "Nobel," not "noble.' They are named for Alfred Nobel, the man who, in 1895, gave the fortune that continues to fund the prizes to this day. Mr. Nobel was a prolific Swedish inventor. His most important invention was dynamite! Gunpowder didn't provide enough explosive force, so mining engineers turned to nitroglycerine, a wonderfully powerful explosive that explodes if the person carrying it trips. Dynamite provided a more convenient and safer alternative, extending the lives of those who carried explosives down into mines. And it made Mr. Nobel rich. RICH!
Bowen studied the crystallization and melting of basaltic lava. Think Hawaiian Islands, Iceland, and any undersea volcano. Basalt is a rock, a mixture of minerals – the so-called rock-forming minerals – but not the minerals most of us collect.

Most minerals melt if you heat them enough. When the melt cools, the mineral will crystalize again at just about the same temperature at which it melted. If you heat solid basalt, different minerals in the basalt will melt at different temperatures. It’s a lot like distilling alcohol from a mixture of alcohol and water. If you heat the mixture, the alcohol boils off first. Catch it, condense it, and drink it, and you won’t worry about the details in the rest of this article.
If magma is cooling, olivine is the first mineral to crystallize. It is denser than the magma, and therefore it sinks to the bottom of the magma chamber.
Bowen put the minerals that typically form from basaltic lava in order by the temperature at which they melt. Part of his genius was to realize that there are two simultaneous sequences. Three sequences if you count the bottom of the Y. Here they are in the standard way of picturing the sequences.
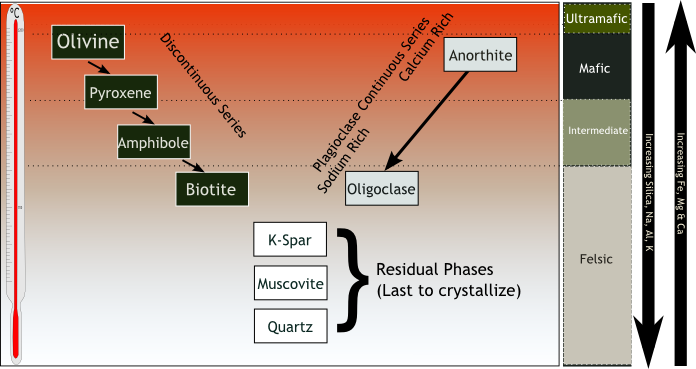
Let’s discuss this diagram. There is a lot in it.
The thermometer on the left has no labels, but the standard labels are 1600° C at the top, where the olivine starts to crystalize, and 800° C at the bottom, where the quartz has all crystalized. Below 800° C, the
magma has all crystalized. The former liquid magma is now solid rock. Between 800°C and 1600° C, the mixture is partly melted and partly solid. A high-temperature slushy.

The left branch of the “Y” is called the discontinuous series. As the magma cools to about 1600° C, olivine forms and settles out. At a somewhat lower temperature, olivine no longer crystalizes. Now we get pyroxene, which settles out on top of the olivine. Then amphibole, and then biotite. Discontinuous refers to the sequence of different minerals.
Now the above description is a little misleading. It sounds as if olivine and pyroxene and the others are flowing around in the magma. Ah – no. The magma includes magnesium, iron, silicate radicals (silicon bonded to oxygen), and other stuff. The bonds between the silicates and the metals form as energy is removed from the magma as it cools. The process is actually a lot more complicated, but this is close enough for our newsletter.
As the minerals crystallize and sink, the percentage of iron and magnesium in the magma decreases. This process, as well as the lower amount of energy available, allows different minerals to form from magma as it cools.
The right-hand branch charts crystallization through the same temperature range, but on this branch, the crystallization is only of a single mineral – plagioclase feldspar. As the temperature decreases, the composition of the plagioclase changes, but it’s all plagioclase. Around 1600° C, the plagioclase is anorthite (calcium plagioclase).
At the bottom of this branch, the plagioclase crystallizing is albite (sodium plagioclase). As the temperature decreases, the percentage of calcium decreases from 100% to 0%, and the percentage of sodium correspondingly increases. Plagioclase can be any percentage of calcium and sodium, as long as the percentages add up to 100%. It’s a little more complicated than that, but since we are about at the limit of my understanding, we’ll let it go.

Since both branches are happening at once, at the high temperatures, the crystals sinking to the bottom are a mixture of olivine and high-calcium plagioclase. Then a mixture of pyroxene and calcium-sodium plagioclase. And so on.
By the time the magma has cooled to where the branches meet, the iron, magnesium, and calcium are essentially gone. What’s left are silicate radicals, sodium, aluminum, and potassium. The potassium feldspar (K-spar), muscovite and quartz form from them.

Other information in the diagram: The arrows on the right show the decreasing iron, magnesium, & calcium and increasing silicate, sodium, aluminum, & potassium. The column just to the left of the arrows introduces a couple of names. Mafic means a mineral that is high in iron and magnesium. Felsic is the opposite: a mineral low in iron and magnesium, thus high in silicate, aluminum, sodium and potassium. Mafic minerals tend to be dense and dark colored. Think gabbro. Felsic minerals are less dense (but they are still rocks) and lighter colored. Think granite.
I think that finishes the description of the diagram of Bowen’s Reaction Series. There’s more to it than that. A universal rule about anything in science is “It’s not that simple.” But we’ll stop here.
Aren’t you glad we distilled that alcohol? Didn’t that make the article easier to get through?

And finally, although Dr. Bowen never won a Nobel Prize, Wikipedia reports that he had a small lunar crater named after him. That is incorrect. The crater was named for Dr. Ira S Bowen, an American astronomer and physicist.

